 
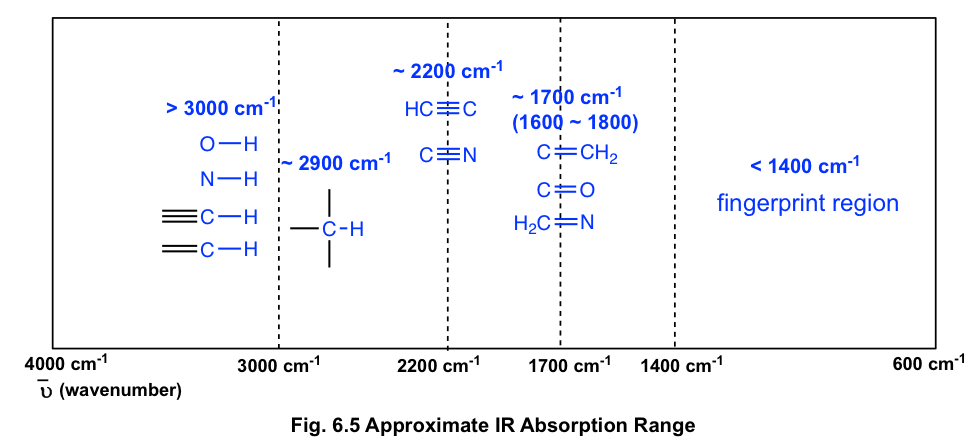
And in complex molecules more than one type of bonds may be present. For strong bonds the frequency of radiation involved will be high and for weak bond, the frequency of radiation involved will be low. One of them is the nature of bonding in molecules. The frequency at which the molecule absorbs the infrared radiation depends on many factors. If the interaction with infrared radiation causes a change in dipole moment of the molecule then it is said to be infrared active. In infrared spectroscopy, the criterion for a molecule to be infrared active is related to its permanent dipole moment. The absorption band in the range of 600 − 1400 c m − 1 1. If two pure samples show an identical infrared spectrum, it can be agreed that they are equivalent or similar compound. Since most organic molecules have such bonds, most organic molecules will display those bands in their spectrum. Of these the most useful are the C-H bands, which appear around 3000 cm-1. Their IR spectrum displays only C-C and C-H bond vibrations. Infrared spectra can also provide information about the molecular fingerprint, which features a unique pattern or region for comparing the respective molecules. IR SPECTRUM OF ALKANES Alkanes have no functional groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
